 It monitors 8650 vaccinated subjects with various pathologies including cancer to evaluate their relative capacity to produce antibodies against SARS-CoV- 2 (the study includes a control group of 1850 subjects without the targeted pathologies). In France, the ANRS S0001S COV-POPART cohort study was recently launched (NCT04824651). The ESMO recently emphasised the importance of monitoring COVID-19 vaccine effects in cancer patients through specific studies and registries. The association of chemotherapy and targeted therapies with CPI treatment in most therapeutic situations also generates potential difficulties in data interpretation, further increasing the need for multi-cohort studies. The immunotherapeutic management of malignant melanoma and lung cancer are clear illustrations of these current developments in CPI treatment. This type of long-term follow-up is particularly necessary when considering current developments in CPI treatment and the increasing role played by their adjuvant setting. Study parameters include the antibody response, the SARS-CoV-2-specific T cell response and the functional and phenotypical characterisation of the cellular immune response. This prospective, multicentric trial aims to closely examine on a long-term basis whether immunotherapy and chemotherapy, alone or combined, influence COVID-19 vaccination in treated patients. This is the main objective of the recently launched VOICE trial. To this end, along with the classical parameters of patient follow-up, more specific immunology-based investigations should be conducted to examine all the aspects of the long-term immune response. express more or less strong messages of caution and point to the need to gain a deeper knowledge of the reciprocal interaction between COVID-19 vaccination and CPI cancer treatment by investigating large cohorts of patients. 
However, the authors recognise the possibility that rare IRAEs could be identified in larger cohorts of patients under COVID-19 vaccination. also mentions the apparent short-term absence of immunologically related adverse events (IRAEs) in a subgroup of 134 patients treated by CPIs who received the BNT162b2 mRNA COVID-19 vaccine. On the other hand, the report by Waissengrin et al. A larger set of patients would certainly be necessary to confirm their findings and deliver a clear message on this point. The authors nevertheless consider their data as supporting the short-term safety of the mRNA COVID-19 vaccine in patients under CPIs. Based on a relatively limited number of cases, the authors note that when compared to matched controls, CPI therapy results in a constant and variable increase of all COVID-19 vaccination side effects, which is cause for alarm. reports on the BNT162b2 messenger RNA (mRNA) COVID-19 vaccine administered in cancer patients under checkpoint inhibitors (CPIs). One recent short article by Waissengrin et al. , especially those in phase III vaccine trials. 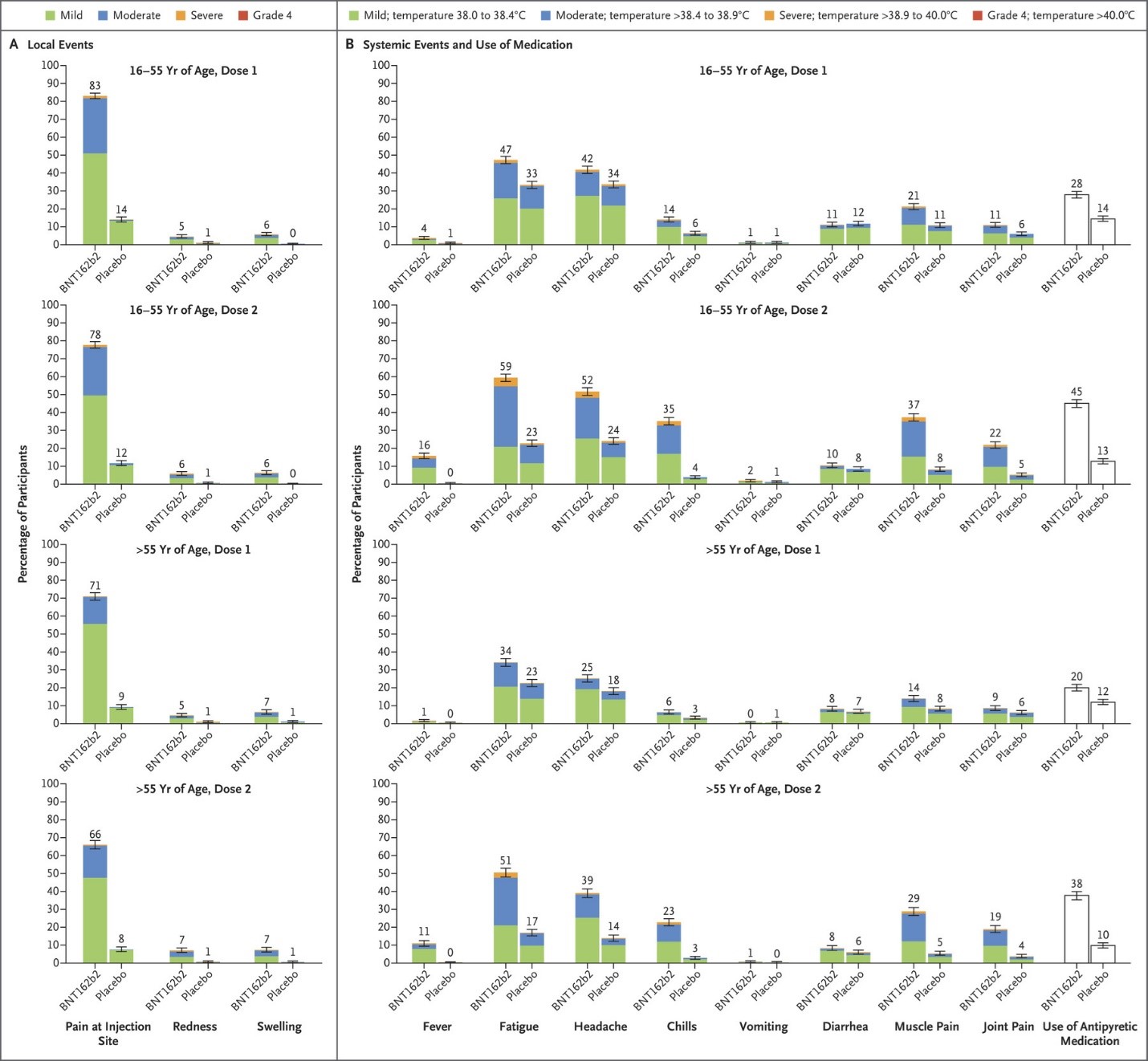
There is a scarcity of data on the consequences of COVID-19 vaccination in cancer patients under specific treatments, as recently stressed by Korompoki et al. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
